Principles of Magnetic Resonance
Magnetic Resonance Imaging (MRI) is based on the behavior of nuclear spins in a strong magnetic field. Atomic nuclei with an odd number of protons or neutrons (like the hydrogen proton in water) possess an intrinsic spin, which gives rise to a magnetic moment. In an external magnetic field , these magnetic moments tend to align with the field, and any that are not perfectly aligned will precess around the field direction at a characteristic Larmor frequency. This is given by the Larmor equation:
where is the angular precession frequency (in radians per second) and is the gyromagnetic ratio (a constant for each nuclear species).
The gyromagnetic ratio determines how fast a given type of nucleus precesses in a magnetic field. For protons (), , meaning at the proton precesses at about 64 MHz and at 3 T about 128 MHz .
Different nuclei have different values. For examples::
- — ,
- — ,
- — .
Among biologically relevant nuclei, the proton has one of the highest gyromagnetic ratios, which is why hydrogen (abundant in water and fat) is predominantly used in MRI.
In essence, a nucleus with spin behaves like a tiny top or bar magnet. When placed in , it will precess analogously to a spinning top in a gravitational field. The precession frequency is proportional to the field strength. This forms the basis for nuclear magnetic resonance: if one applies radiofrequency (RF) energy at the Larmor frequency, one can resonantly excite the spins. In MRI, the main static field is very strong (typically 1.5 T, 3 T, or higher in research scanners), setting the stage for all protons to precess in unison at a well-defined frequency.
Spin Excitation and Relaxation (T1 and T2)
In thermal equilibrium, slightly more spins align with (low energy state) than against it, yielding a net longitudinal magnetization along the field. An RF pulse (usually a 90° pulse) at the Larmor frequency tips some of this magnetization into the transverse plane, creating transverse magnetization .
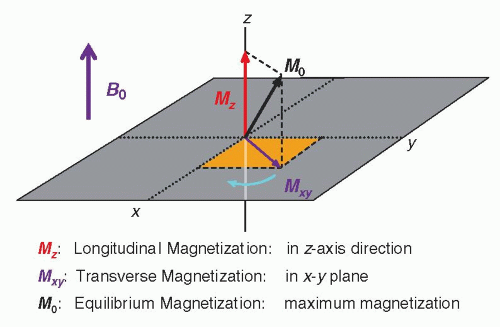 (Image from radiologykey.com)
(Image from radiologykey.com)
Once the RF pulse is turned off, the spins begin to return to equilibrium through two independent exponential processes known as relaxation:
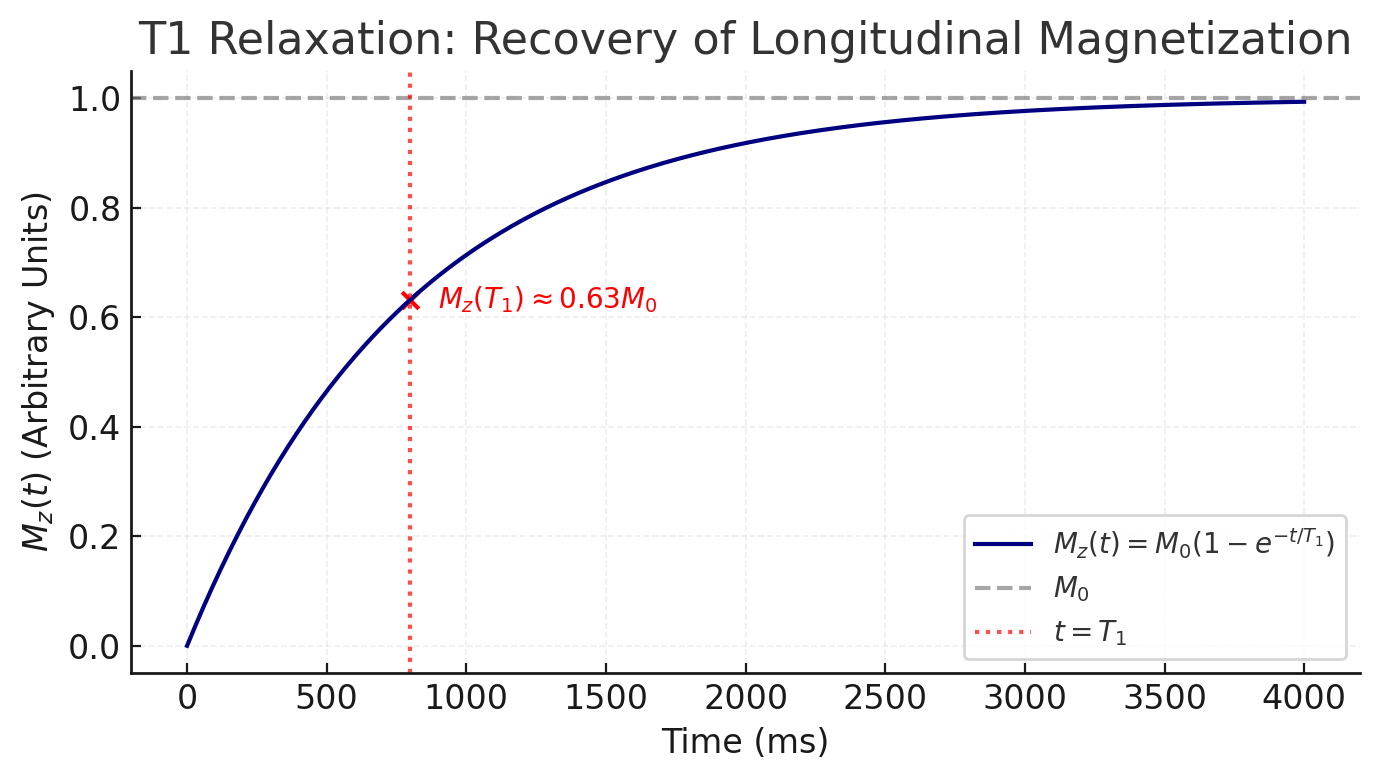
- T1 relaxation (longitudinal or spin-lattice relaxation): This is the recovery of back to its equilibrium value along the z-axis (parallel to ). Physically, the spin system releases energy to the surrounding lattice (the molecular environment) as spins realign with the field. The regrowth of follows an exponential recovery:
where is the time constant for 63% recovery of the longitudinal magnetization . Each tissue has a characteristic (ranging from hundreds of milliseconds to seconds in biological tissues), and is generally longer at higher field strengths. Intuitively, depends on how efficiently a nucleus can transfer energy to its surroundings; typically, protons in water have long (on the order of 2–3 s at 1.5 T), whereas protons in fat or protein have shorter (hundreds of ms or less) because of more efficient energy exchange with the lattice.
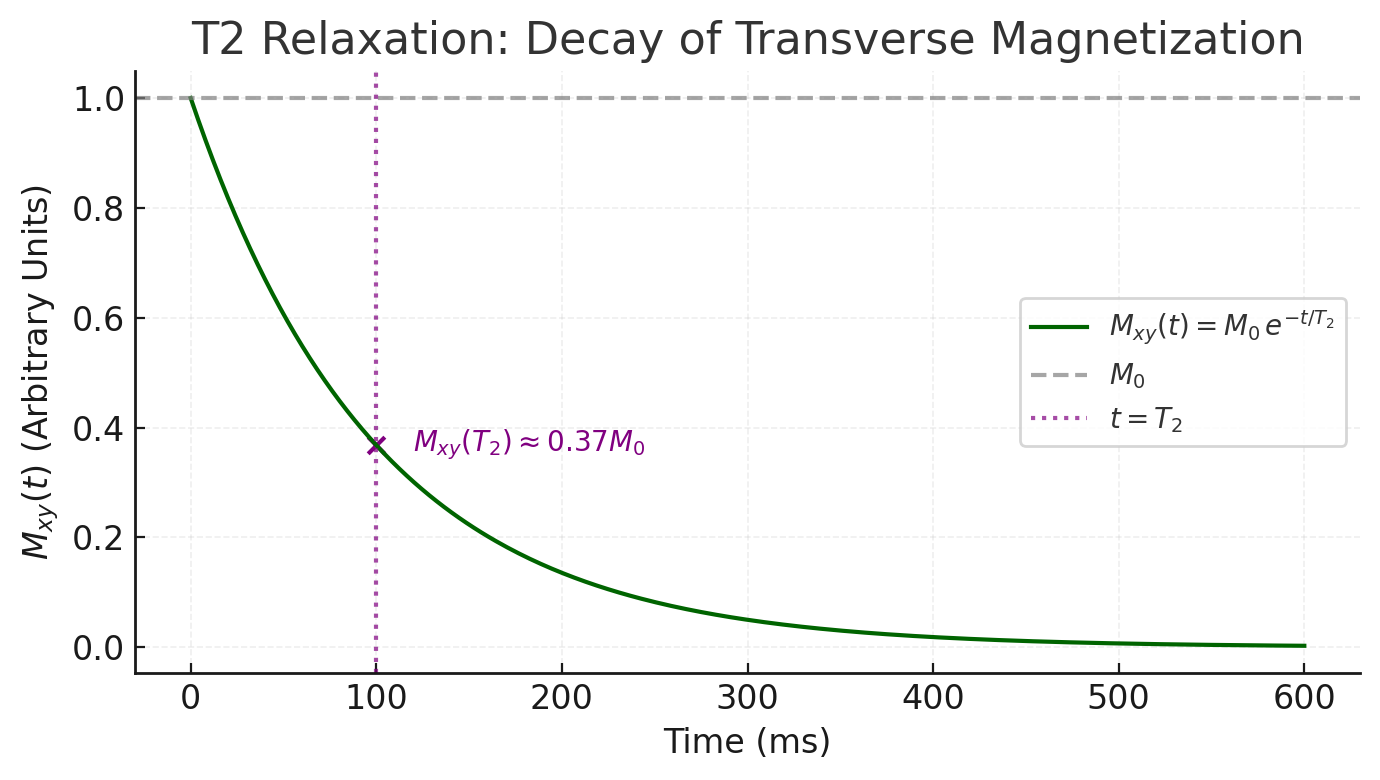
- T2 relaxation (transverse or spin-spin relaxation): This is the decay of in the transverse plane due to dephasing of individual spins. Immediately after an RF pulse, the spins are momentarily in phase, but various interactions cause them to fall out of phase with each other over time, leading to a loss of net transverse magnetization. The decay of is exponential:
where is the time constant for the transverse magnetization to decay to 37% of its initial value. is typically shorter than for a given tissue (often by a factor of 5–10) . This is because many microscopic processes (like random spin-spin interactions and local field inhomogeneities) cause dephasing without energy loss, speeding up the decay. Pure liquids (like cerebrospinal fluid or water) have relatively long (hundreds of milliseconds), whereas solids or tightly bound protons have very short (tens of milliseconds or less) due to more rapid dephasing .
Importantly, there is also (T2-star) relaxation, which is the effective transverse relaxation that includes not only the spin-spin interactions (true decay) but also dephasing from static magnetic field inhomogeneities. In a perfectly homogeneous field, transverse decay would be governed by alone. In a real MRI scanner, however, slight field non-uniformities and susceptibility variations cause additional dephasing, making the observed decay faster (with a time constant ). is particularly important for gradient-echo MRI sequences and for BOLD fMRI, as we will see later. A spin-echo sequence can refocus static inhomogeneity effects (recovering decay), whereas gradient-echo sequences do not, thus capturing effects.
MRI Contrast Mechanisms (T1- vs T2-Weighted Imaging)
MRI scanners can be “tuned” to produce images with different contrasts by exploiting the differences in , , and proton density of tissues. The timing parameters of a pulse sequence — primarily the repetition time (TR) and echo time (TE) — determine how the image contrast is weighted.
Repetition time (TR) is the time interval between the start of one RF excitation pulse and the start of the next in an MRI pulse sequence. TR determines how much longitudinal magnetization (along ) has recovered before the next excitation.
Echo time (TE) is the time between the application of the RF excitation pulse and the moment the MR signal (the “echo”) is measured. TE controls how much transverse magnetization decay (T2 or T2*) has occurred before signal acquisition.
- T1-weighted images
Use a short TR and short TE. A short TR (on the order of the tissue or less, e.g. ~300–600 ms) means tissues haven’t fully recovered their before the next excitation, so tissues with shorter recover more and appear brighter, while long tissues remain darker. A short TE (e.g. < 30 ms) minimizes decay effects, so contrast is mainly due to differences.
Example: Fat (short ~240 ms) appears bright on T1-weighted images, whereas fluid like cerebrospinal fluid (long ~3000 ms) appears dark. T1-weighted scans provide clear anatomical detail and are often used post-contrast, because gadolinium-based contrast agents shorten dramatically, making enhancing tissues light up on T1 images.
- T2-weighted images
Use a long TR and long TE. A long TR (≫ , e.g. > 2000 ms) allows full or near-full recovery in all tissues before the next excitation, so differences are minimized. A long TE (e.g. 70–100+ ms) allows substantial decay, so tissues with longer will retain more signal and appear brighter, while short tissues lose signal and appear dark.
Example: Fluid (long ~3000 ms for CSF) remains bright on T2-weighted images (since it decays slowly), whereas tissues like white matter (shorter ~90 ms) lose most of their transverse magnetization by the echo time and appear relatively dark. T2-weighted scans are very sensitive to pathology because many lesions (edema, inflammation) have higher water content and thus prolonged , making them stand out bright against darker normal tissue.
- Proton-density (PD) weighted images:
Use a long TR (to minimize differences) and a short TE (to minimize differences). This yields contrast primarily based on the number of protons (hydrogen density) in each tissue. PD-weighted images are less common in the brain but are sometimes used in orthopedic MRI.
(Note: Many MRI sequences exist beyond simple spin-echo. For example, FLAIR is a T2-weighted sequence with fluid signal suppressed, useful in brain imaging to see periventricular lesions; gradient-echo sequences can be T2-weighted, etc. However, the basic principles of and contrast apply across these variations.)
References and Further Reading
- Buxton RB. Introduction to Functional Magnetic Resonance Imaging: Principles and Techniques. Cambridge Univ. Press, 2009. (Comprehensive text on fMRI physics and physiology)
- [MRIQuestions.com](An excellent website with clear explanations)